COVID-19 Is Inspiring Innovation
By Aranka Anema
Advancements are sparked from new collaborations in science and business during the pandemic.
May 8, 2020
The Long View Medicine Technology Cartography Social Science
Editors' Note: Aranka Anema has previously written about using data from the internet and social media to create a tracking system for infectious diseases. Here, she updates that work by describing new technology and advances in data science.
Ingenuity is born from adversity, as the saying goes. And 2020 has already had plenty of adversity for families, health care workers, public health officials, and those who now find themselves in isolation at home. Most businesses have also suffered tremendous losses that will have long-term repercussions because of major drops in consumer spending and shortfalls in federal aid.
Could there be any silver lining to this crisis? Perhaps this: We have been forced to accelerate the adoption of open innovation, which has allowed us to better deal with this crisis and may make us more resilient and proactive in future public health emergencies. In this case, open innovation refers to collaboration that happens within and among different institutions to produce a distributed innovation process for the cocreation of products and services. Companies, government, nonprofits, and citizens are collaborating in unprecedented ways to develop low-cost and efficient tools for the prevention, detection, and treatment of COVID-19. These initiatives have been born out of a common spirit of social responsibility and humanitarianism, and they position companies and governments to be more resilient and proactive in future public health emergencies. There are five key areas in which open innovation has proliferated.
Data Sharing
To be effective, public health responses to epidemics must be based on data. In the past, gaps in data have impeded epidemic surveillance, forecasting, and interventions, notably during the 2014 Ebola epidemic in West Africa. With the rise of COVID-19, many organizations expeditiously mobilized to increase access to timely and open data. These organizations include the UN Office for the Coordination of Humanitarian Affairs’ Humanitarian Data Exchange, which hosts several COVID-19 data-sharing initiatives, including the United Nations COVID-19 Data Hub, where statisticians can share guidance, best practices, and information resources. There’s also the Johns Hopkins University Novel Coronavirus COVID-19 Data Repository, which maintains country-specific incidence and mortality statistics compiled from governments globally, and the COVID-19 Testing Insights Initiative, which shares state-specific testing data.
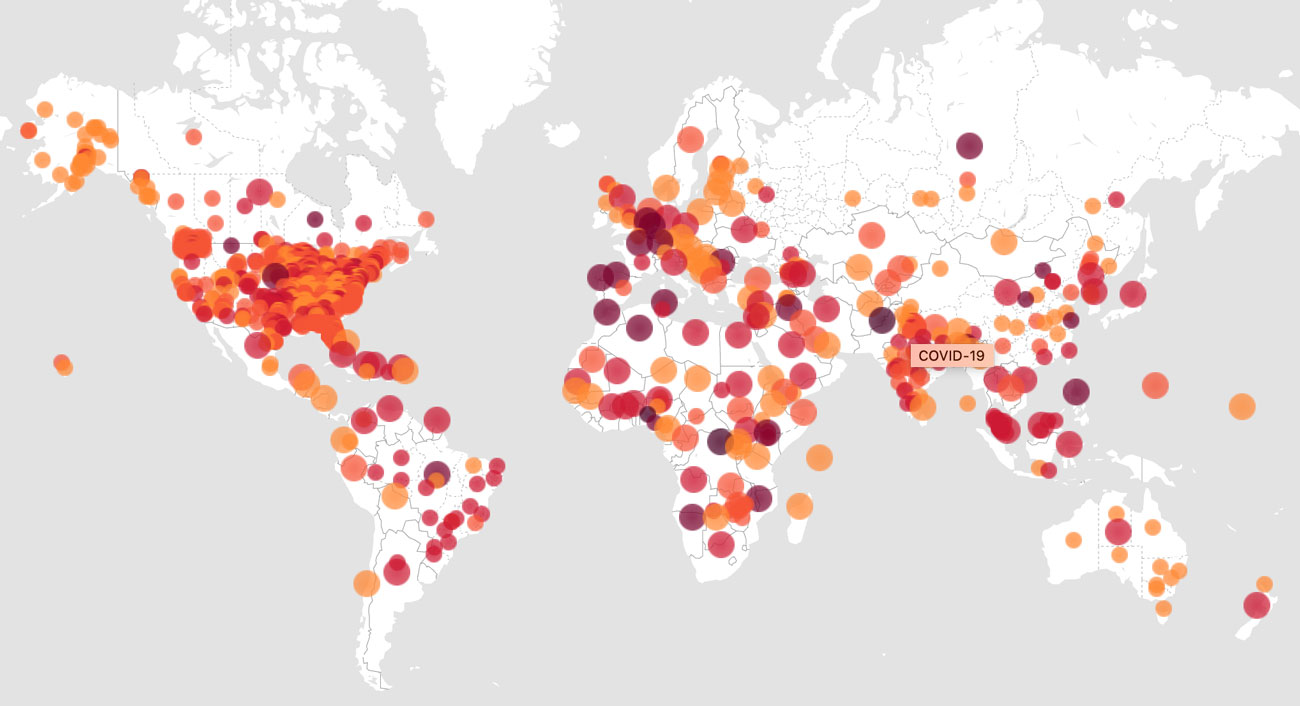
HealthMap brings together disparate data sources, including online news aggregators, eyewitness reports, expert-curated discussions and validated official reports, to create a unified and comprehensive view of COVID-19 cases and deaths (shown above). The COVID-19 Open Research Dataset (CORD-19) has freely accessible, machine-readable scientific articles for artificial intelligence analyses. Platforms such as Nextstrain provide open-sourced pathogen genome data along with analytic and visualization tools for public use, allowing scientists to trace and monitor the COVID-19 pandemic faster than any previous outbreak. In addition, the bioRxiv repository of preprint research papers releases torrents of data on a daily basis (although preprints should not be viewed as equivalent to papers that have passed peer review). These open initiatives and many others have demonstrated an overwhelming commitment to cocreation and solution-finding for the common good during COVID-19 and have set a precedent for ongoing and future infectious diseases data sharing.
Modeling
Over the past three months, models using varying statistical and mathematical techniques have aimed to project the global number of COVID-19 cases and deaths, and the effect of public health interventions. A recent systematic review of projection models specific to COVID-19 identified 31 models to which the authors ascribed good to excellent performance—but all of the models were found to contain bias that reduced their reliability (see more about careful interpretation of epidemic models here). These projections, although imperfect, are essential for guiding public health policies and responses intended to curb the pandemic. Some notable examples of COVID-19 models include the SEIR (Susceptible, Exposed, Infectious, Recovered) models developed by Columbia University and the Massachusetts Institute of Technology; mechanistic transmission models by Imperial College London; a model developed by Los Alamos National Laboratory (shown below), and one created by the University of Washington’s Institute for Health Metrics and Evaluation.
New partnerships are aiming to produce models with increasing accuracy. One example is ImmunityBio’s collaboration with Microsoft Azure, which aims to enhance computing capability for researchers who are studying COVID-19 biochemistry. Meanwhile, institutions such as the United Kingdom’s National Health Service have provided guidelines to data scientists on how to make new source code open and reusable, and in machine-readable file formats, to eliminate inconsistencies in data quality. The proliferation of models during these past few months, and the openness of data associated with them, can enable the development of prospective and retrospective studies of COVID-19 that can improve pandemic responses.
Biotech R&D
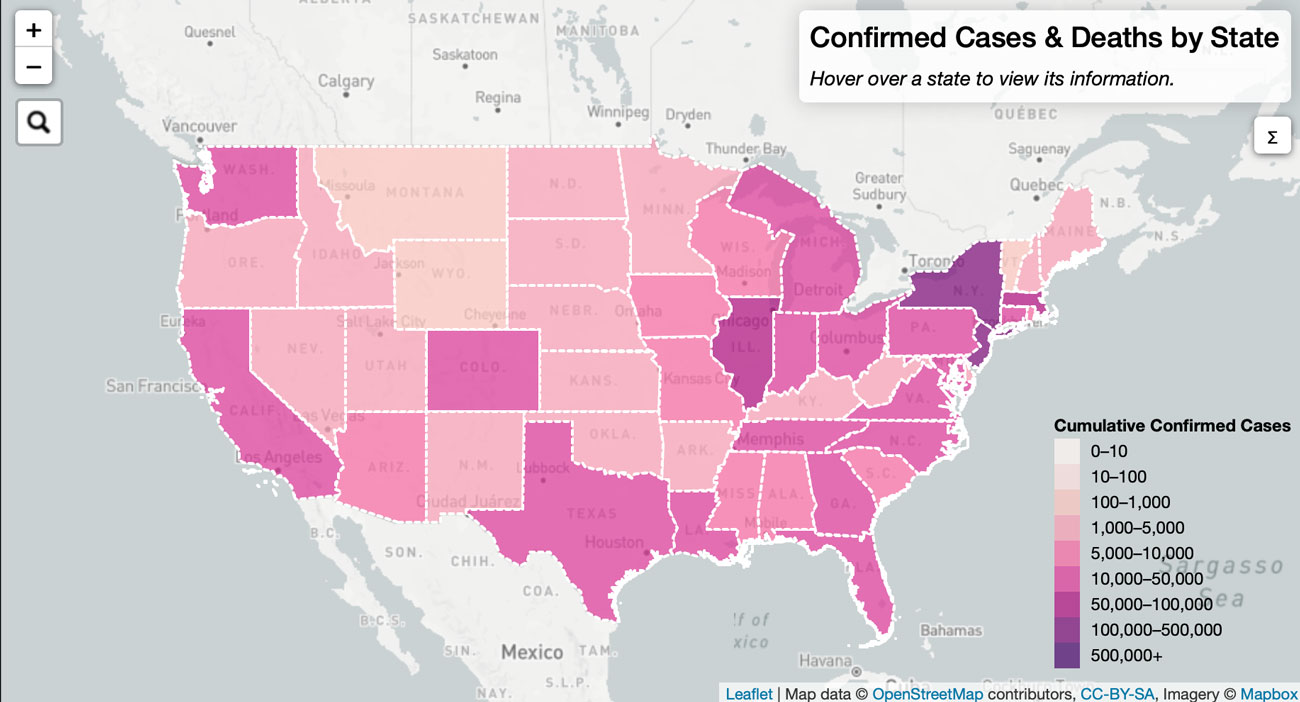
The timeline for drug and vaccine development traditionally takes 10 to 15 years from prediscovery (during which scientists conduct research to understand cellular and molecular processes and pathways) to availability on pharmacy shelves. Each year, a couple of dozen new products are licensed by the U.S. Food and Drug Administration. However, in their wake lie thousands of candidates that didn’t survive because of lack of funding, inability to demonstrate efficacy and safety, or poor participant recruitment, enrollment, and retention. Currently, there are over 1,000 vaccine and therapeutic trials ongoing around the world (mapped below), but only a small fraction will ever deliver a viable product.
Collaboration between established biotech firms can significantly cut research and development timelines. The recently announced open innovation collaboration between Sanofi and GlaxoSmithKline (GSK) constitutes one of the largest manufacturing partnerships formed to fight COVID-19. Sanofi is bringing its S-protein COVID-19 antigen (an immune system signal that attaches to a surface protein on SARS-CoV-2) to the table and GSK is contributing its vaccine adjuvant technology (which allows a vaccine to use a smaller amount of protein per dose). AstraZenica and Oxford University have similarly partnered to develop and globally distribute a recombinant adenovirus vaccine against COVID-19. Public-private partnerships, such as those led by the U.S. National Institutes of Health, aim to standardize research methods across trials and to prioritize near-term vaccine and therapeutic candidates.
Exponential Technologies
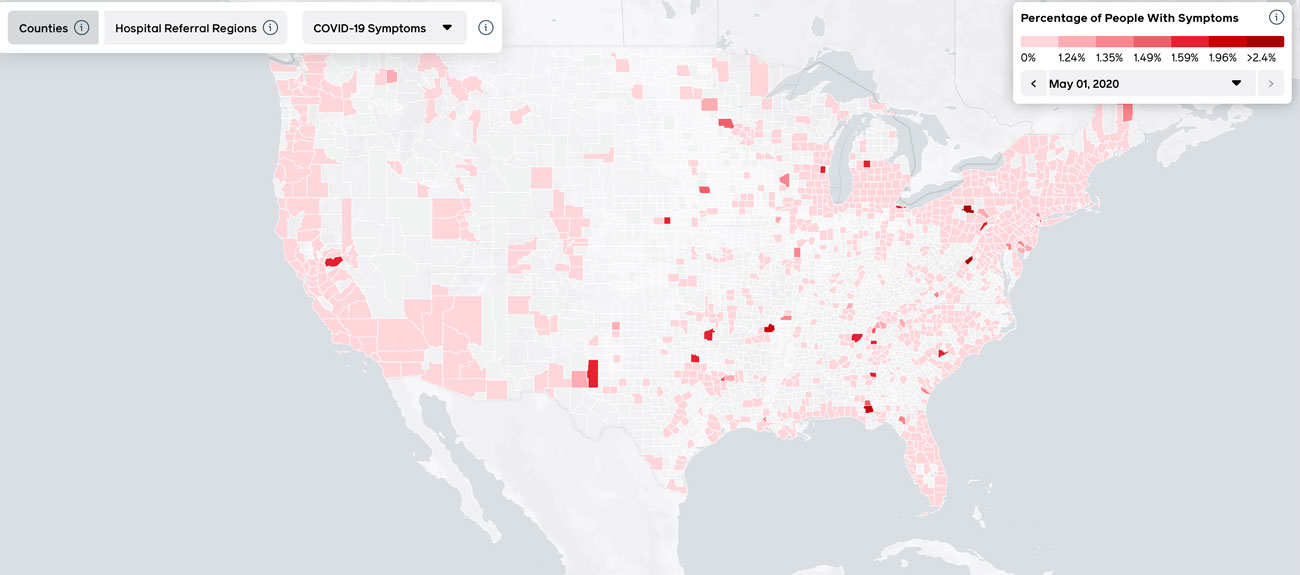
The COVID-19 pandemic has also accelerated the use of what are called exponential technologies, ones such as 3D printing, in which the capability has risen quickly while its costs have dropped. Shoe manufacturers, including Nike, Adidas, and New Balance, have all repurposed their 3D printing capabilities to make high-volume, low-cost personal protective equipment (PPE) for health care workers. The NIH 3D Printer Exchange has published a repository of PPE designs that have undergone review in a clinical setting. HP and partners have made their 3D design files freely available. Maker spaces, such as Maker Mask and ArtistsAsylum, are using open source designs and encouraging citizen scientists to make their own masks. Meanwhile, automobile manufacturers such as Tesla (whose prototype is shown below), General Motors, Volkswagen, and Ford are partnering with government agencies and retooling their 3D printers to create ventilators for distribution to hospitals across the United States.

Image courtesy of Tesla
Autonomous vehicles and robotics are other exponential technologies that have played a major role in the COVID-19 response. Numerous countries, including China and Ghana, have successfully used drones to disseminate information, testing equipment, medical supplies, and food to rural and urban areas. Collaborations between health care organizations and self-driving car manufacturers, such as that between Mayo Clinic and Beep, have made it possible to limit human exposure to the virus during the transportation of COVID-19 test samples. Driverless delivery robots, such as Nuro R2, are being used for low-contact delivery of groceries and other essentials. Robots have been used for disinfection and to enable virtual health care. Although numerous regulatory hurdles exist for broad roll out of health care robots and autonomous vehicles, companies and governments have leveraged COVID-19 as an opportunity to expand testing and data collection in an effort to create evidence-based regulations for future responses to health emergencies.
Citizen Science
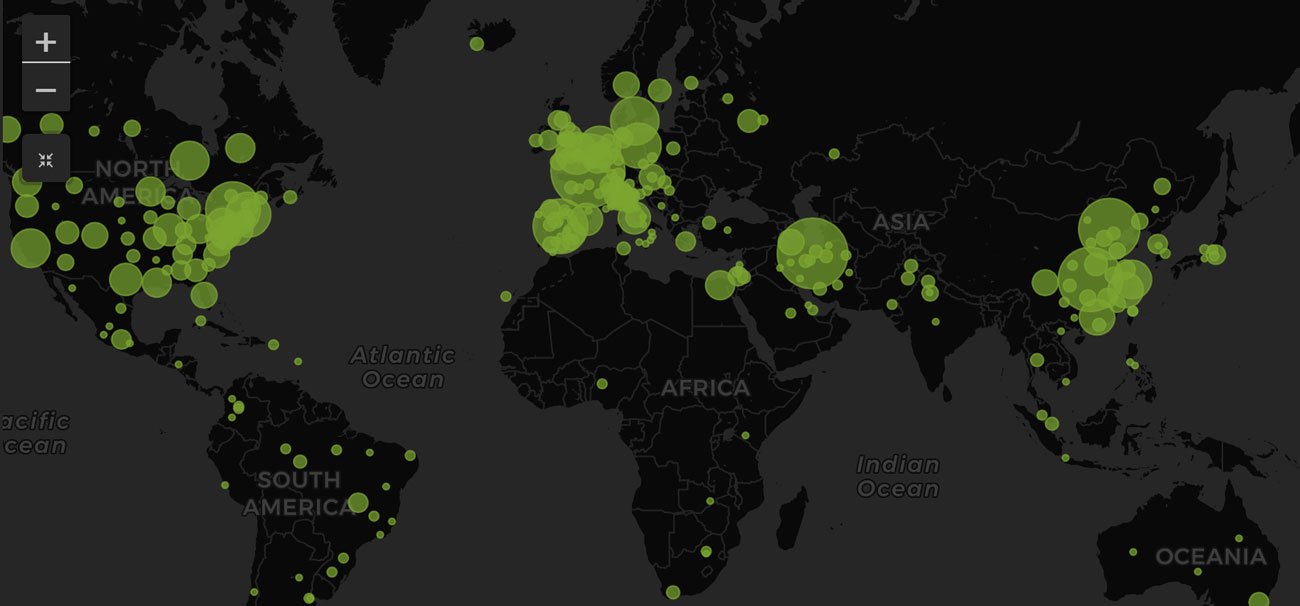
Citizen science is a rapidly growing paradigm of research whereby members of the general public engage in informal empirical research. Numerous resources have emerged for citizen scientists who wish to contribute to the COVID-19 response, ranging from collective group efforts to innovations by individuals. Facebook and Carnegie Mellon University recently published a map estimating the number of COVID-19 cases across the United States based on self-reporting of the disease by consenting social media users (shown below).
Crowdsourcing projects such as the Paris-based Just One Giant Lab (JOGL) Open COVID-19 Initiative are convening citizen scientists to build low-cost, open-source prevention, diagnostic, and therapeutic tools. The “quantified self” movement, which advocates personal data collection about oneself to aid in one’s own health optimization, has also mobilized; quantified self blogs have shared stories about how to use wearable devices to provide early warnings of COVID-19 infection. One citizen scientist recently developed a COVID-19 antibody testing kit, which is now undergoing formal study with support from Oregon State University. These and many other citizen science initiatives during COVID-19 have brought attention and, to a certain extent, legitimacy to a new paradigm of research that allows everyday people to meaningfully contribute their time and knowledge.
Laying a Path for Innovation
COVID-19 has posed tremendous challenges to global health. However, in its wake we will find it has also stimulated tremendous innovation in areas of data sharing, modeling, biotech, exponential technologies, and citizen science. Many new insights and capabilities derived from COVID-19 projects will continue to evolve over the coming years, allowing us to be more proactive and prepared for future public health emergencies. 2020 will be remembered as an inflection point, creating a path of open innovation towards the common good.
3 June 2020 Editor’s Note: This post was updated with a new map and caption to reflect the growing number of vaccine trials, as well as updated by replacing one image and adding a sentence to highlight HealthMap, a project the author works on.
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.